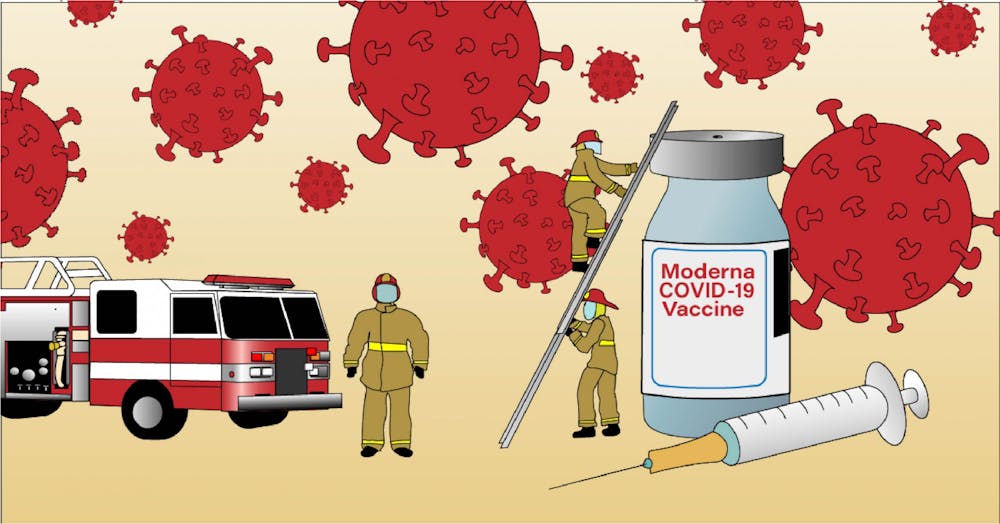
In August, about 10 members of the Gainesville Fire Rescue team volunteered to become a few of the 30,000 nationwide participants in Moderna’s Phase 3 COVE vaccine trial.
For the past six months, GFR has worked with ENCORE Research Group at the Jacksonville Center for Clinical Research.
The select firefighters who participated in the vaccine trial were allowed to share the application with their families and friends. Chief David Sutton, District Chief of Training at GFR, was able to include his wife and family friend to participate in the trial.
“I felt a moral obligation to help,” Chief Sutton said. “We were definitely looking to contribute to making the vaccine a successful initiative, but we also wanted to try to protect ourselves and our families.”
The process of participating in the trial began with the two vaccine shots, ENCORE Vice President of Recruitment Sharon Smith said. The firefighters received their first injection in late August and early September, then they received the second a month later. ENCORE monitored the firefighters after each shot, including frequent calls to monitor any COVID-19 symptoms. The participants were “blinded,” meaning they do not know if they received the vaccine or a placebo, Smith added.
The researchers were seeking volunteers at higher risk of contracting COVID-19, such as first responders, healthcare workers and minorities, Smith said. Over 500 people contacted ENCORE about participating in the trial, including GFR Assistant Chief Shawn Hillhouse.
Smith said Hillhouse asked if they were administering the trial and if he could enroll some of his firefighters. He referred 30 firefighters from Gainesville, but only 10 of them were able to enroll in the study.
“We have a relatively healthy workforce that can participate in something like this without worrying about minor symptoms that are involved,” Chief Hillhouse said.
Both chiefs felt confident in the process of the trial. ENCORE informed them about the procedure, and they researched the vaccine and its side effects before the study.
Both Chief Sutton and Chief Hillhouse requested to be unblinded by the research group so that they could decide whether or not to take the vaccine with the rest of their team. By the time the Alachua County Health Department shipped the Moderna COVID-19 vaccine to GFR on Dec. 30, the firefighters in the trial had received both of their injections.
Sutton says it was an added bonus when his trial turned out to be the vaccine instead of the placebo.
“So, whereas, most people got it in January, I received the benefit of having the vaccine in August, because I participated in the trial,” Sutton said.
Another feature of the trial is the Moderna-sponsored patient stipend that covers the time and traveling expense of each volunteer, Smith said, adding that ENCORE sends a check in the mail to the participant within two weeks of each visit or phone call. By the time a participant completes the study, they should have accumulated $1,872 in reimbursements from Moderna.
Once Moderna reached its quota of 30,000 volunteers in October, ENCORE stopped enrolling volunteers into their trial, Smith said. However, the researchers will continue to follow up with the volunteers for 25 months to collect data about the effectiveness of the vaccine.
“I have another visit scheduled in April,” Sutton said. “But, mostly it's just been filling out the information on the app and receiving a phone call.”
ENCORE is thankful for the firefighters who will continue to donate their time to this trial for the next two years, Smith said. By volunteering with ENCORE, they proved their commitment to serve local and global communities and promote education of the COVID-19 virus.
Contact Jiselle Lee at jlee@alligator.org. Follow her on Twitter @jiselle_lee.
Jiselle Lee was The Alligator’s Summer 2023 Editor-In-Chief. She was previously a reporter with NextShark News and a reporting intern at The Bradenton Herald.