Drinking mucus developed by UF researchers could reshape how inflammatory bowel disease and similar gastrointestinal disorders are treated.
Researchers Luiz Roesch and Brent Sumerlin designed a synthetic mucus in UF laboratories that could treat the disorders directly, rather than just making the condition more manageable.
Inflammatory bowel disease, commonly referred to as IBD, is an umbrella term for chronic disorders caused by inflammation in the digestive tract. The most common are Crohn’s disease and ulcerative colitis. According to the Centers for Disease Control and Prevention, between 2.4 million and 3.4 million Americans suffer from IBD.
Treatments vary but include dietary changes, immunosuppressant drugs, anti-inflammatory medications, antibiotics and surgeries to remove damaged parts of the gastrointestinal tract.
In 2018, the total U.S. health care costs for treating IBD were an estimated $8.5 billion. On average, immunosuppressant drugs alone cost between $10,000 and $14,000 per year. Anti-inflammatory diets also tend to cost about $550 more annually, according to the Harvard School of Public Health.
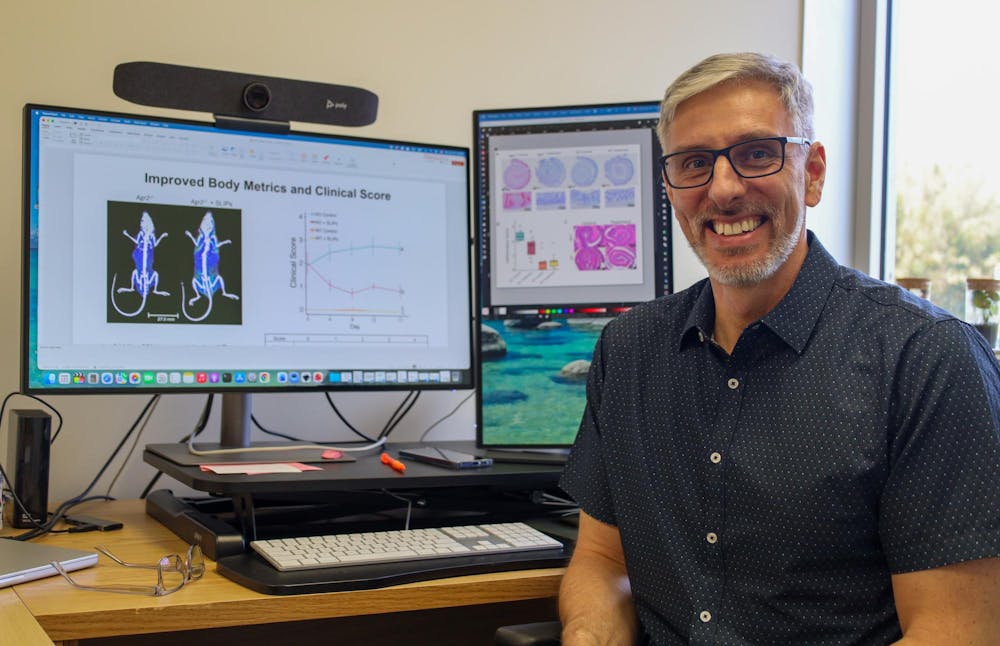
Roesch, one of the researchers developing the synthetic mucus treatment, said existing treatments only focus on relieving symptoms once they appear. That's where the mucus differs.
“When you have too much immune response, a long-term inflammatory process that causes cell damage, that's not beneficial,” Roesch said. “We just want to prevent inflammation from happening in the first place.”
Roesch and his colleague, Sumerlin, worked to mimic the function of natural mucus in the body. Mucus acts as the first line of defense against pathogens through its role as the protective boundary between the intestines and underlying tissues. Damage to the mucus layer leads to gastrointestinal issues. This damage results from underlying genetic issues, smoking or poor diet.
To test the safety and effectiveness of the synthetic mucus, the researchers tested the substance on adult mice for 10 days. They added it to the mice's drinking water and watched for any signs of health issues within the time period.
The mice didn’t experience harmful effects at any dosage. Results showed the substance was able to restore the mucus barrier integrity and mitigate gut inflammation.
Patients suffering from IBD would be able to incorporate the flavorless mucus into their everyday drinking water rather than taking heavy prescriptions or following restrictive diets.
Amanda Ojeda, a UF doctoral candidate working with Roesch, is researching the use of synthetic mucus in reproductive health.
Ojeda envisions the substance to be similar to powdered Gatorade, she said, but for your gut. Patients would drink the “mucinade” daily, and it would eliminate inflammation, which would in turn relieve symptoms.
“Think of it almost as a Band-Aid,” she said. “You want your underlying skin to be protected from the external environment.”
Mandy Dempsey, the senior director of national advancement and volunteer engagement at the Crohn's and Colitis Foundation, said four in 10 patients are responding to existing IBD medications or will lose response over time. She added that advancement in treatments helps patients not only manage the disorder, but also feel less shame or embarrassment. The disease is very stigmatized, she said.
“Obviously, it's not a sexy disease at all, and it's not something that is really talked about as much as it should be,” Dempsey said. “Research is really everything for them. It really provides hope.”
Researchers are currently looking to obtain approval from the Food and Drug Administration; however, there is no clear timeline for public release.
Grace Galloway is a contributing writer for The Alligator.
Grace Galloway is a contributing writer at The Alligator.